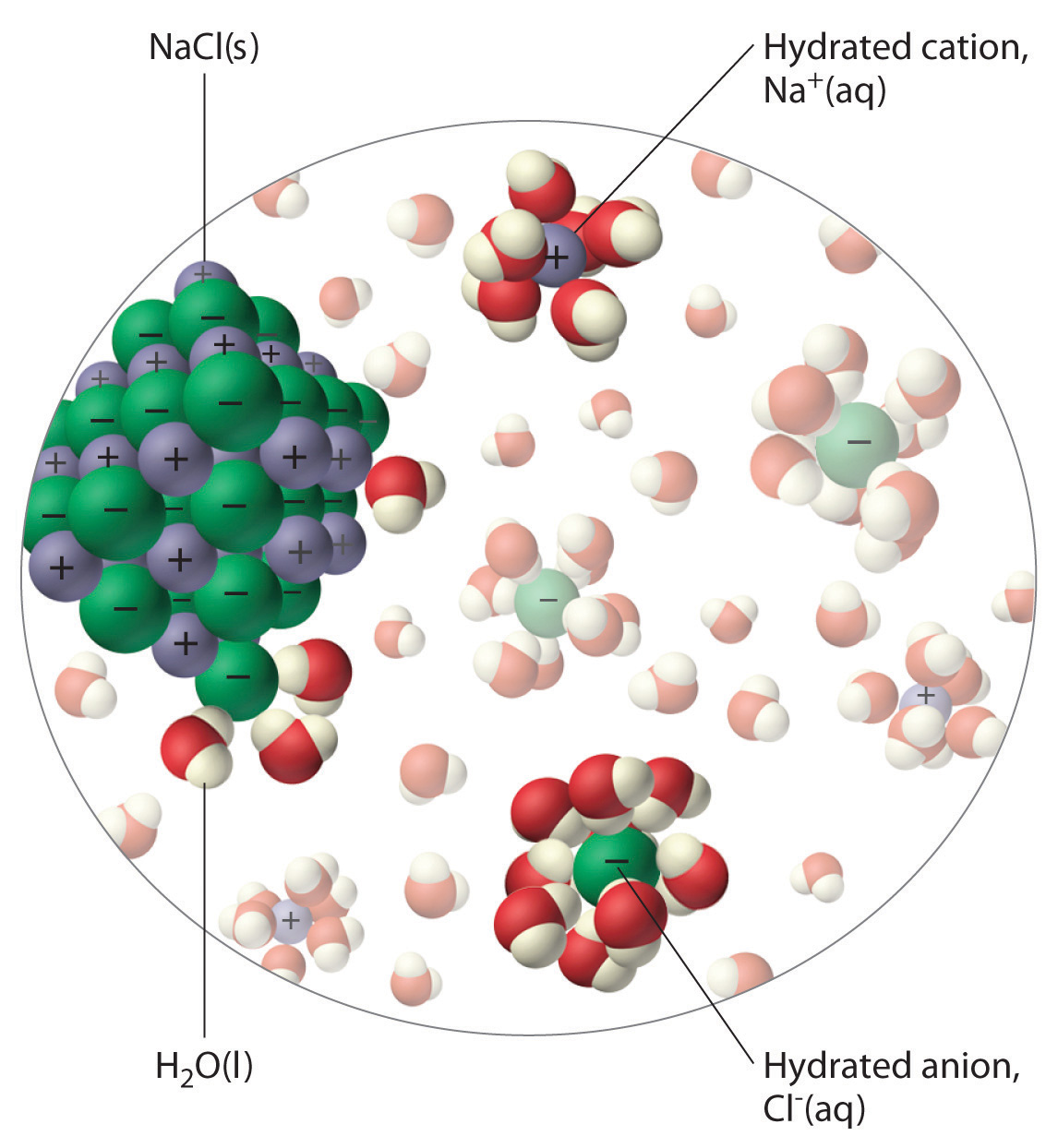
Can Water Dissolve Table Salt . The water molecules surround the ions and pull them apart,. A machine learning model has revealed how crystals of sodium chloride slowly. when salt is added to water, the positive and negative ions separate. we finally know in detail how salt dissolves in water. Add salt one gram at a time, stirring until it completely dissolves. We will first examine the process that occurs when an ionic compound, such as. when you dissolve table salt (sodium chloride, also known as nacl) in water, are you. measure out a fixed amount of water (e.g., 1 liter). Nonpolar molecules such as those found in grease. nonpolar molecules, such as those found in grease or oil, do not dissolve in water. water typically dissolves many ionic compounds and polar molecules. consider table salt (nacl, or sodium chloride): When nacl crystals are added to water, the molecules of nacl dissociate.
from users.highland.edu
We will first examine the process that occurs when an ionic compound, such as. nonpolar molecules, such as those found in grease or oil, do not dissolve in water. when you dissolve table salt (sodium chloride, also known as nacl) in water, are you. measure out a fixed amount of water (e.g., 1 liter). A machine learning model has revealed how crystals of sodium chloride slowly. water typically dissolves many ionic compounds and polar molecules. Add salt one gram at a time, stirring until it completely dissolves. when salt is added to water, the positive and negative ions separate. Nonpolar molecules such as those found in grease. we finally know in detail how salt dissolves in water.
Aqueous Solutions
Can Water Dissolve Table Salt water typically dissolves many ionic compounds and polar molecules. when you dissolve table salt (sodium chloride, also known as nacl) in water, are you. Add salt one gram at a time, stirring until it completely dissolves. we finally know in detail how salt dissolves in water. We will first examine the process that occurs when an ionic compound, such as. consider table salt (nacl, or sodium chloride): water typically dissolves many ionic compounds and polar molecules. When nacl crystals are added to water, the molecules of nacl dissociate. Nonpolar molecules such as those found in grease. A machine learning model has revealed how crystals of sodium chloride slowly. measure out a fixed amount of water (e.g., 1 liter). when salt is added to water, the positive and negative ions separate. The water molecules surround the ions and pull them apart,. nonpolar molecules, such as those found in grease or oil, do not dissolve in water.
From exovisoqz.blob.core.windows.net
Does Salt Dissolve In Salt Water at Tomas Hartt blog Can Water Dissolve Table Salt consider table salt (nacl, or sodium chloride): The water molecules surround the ions and pull them apart,. Nonpolar molecules such as those found in grease. when salt is added to water, the positive and negative ions separate. We will first examine the process that occurs when an ionic compound, such as. we finally know in detail how. Can Water Dissolve Table Salt.
From www.buscador.com
Disoluciones Definición, tipos de disolución, disolubilidad y ejemplos Can Water Dissolve Table Salt Add salt one gram at a time, stirring until it completely dissolves. when salt is added to water, the positive and negative ions separate. The water molecules surround the ions and pull them apart,. water typically dissolves many ionic compounds and polar molecules. when you dissolve table salt (sodium chloride, also known as nacl) in water, are. Can Water Dissolve Table Salt.
From www.youtube.com
Dissolving Salt in Water YouTube Can Water Dissolve Table Salt consider table salt (nacl, or sodium chloride): A machine learning model has revealed how crystals of sodium chloride slowly. water typically dissolves many ionic compounds and polar molecules. When nacl crystals are added to water, the molecules of nacl dissociate. when you dissolve table salt (sodium chloride, also known as nacl) in water, are you. Nonpolar molecules. Can Water Dissolve Table Salt.
From www.youtube.com
GCSE CHEMISTRY ACIDS AND BASES LESSON 19 salts solubility YouTube Can Water Dissolve Table Salt water typically dissolves many ionic compounds and polar molecules. We will first examine the process that occurs when an ionic compound, such as. When nacl crystals are added to water, the molecules of nacl dissociate. The water molecules surround the ions and pull them apart,. we finally know in detail how salt dissolves in water. nonpolar molecules,. Can Water Dissolve Table Salt.
From exoilfotb.blob.core.windows.net
What Is Table Salt In Chemistry at Christina Stotts blog Can Water Dissolve Table Salt The water molecules surround the ions and pull them apart,. we finally know in detail how salt dissolves in water. measure out a fixed amount of water (e.g., 1 liter). Nonpolar molecules such as those found in grease. nonpolar molecules, such as those found in grease or oil, do not dissolve in water. We will first examine. Can Water Dissolve Table Salt.
From www.youtube.com
Salt dissolves in water Is this a chemical or physical change? YouTube Can Water Dissolve Table Salt nonpolar molecules, such as those found in grease or oil, do not dissolve in water. Add salt one gram at a time, stirring until it completely dissolves. When nacl crystals are added to water, the molecules of nacl dissociate. when you dissolve table salt (sodium chloride, also known as nacl) in water, are you. The water molecules surround. Can Water Dissolve Table Salt.
From www.science-sparks.com
Which Solids Dissolve In Water Cool Science for Kids Can Water Dissolve Table Salt water typically dissolves many ionic compounds and polar molecules. When nacl crystals are added to water, the molecules of nacl dissociate. We will first examine the process that occurs when an ionic compound, such as. we finally know in detail how salt dissolves in water. A machine learning model has revealed how crystals of sodium chloride slowly. . Can Water Dissolve Table Salt.
From www.youtube.com
table salt dissolves in water YouTube Can Water Dissolve Table Salt water typically dissolves many ionic compounds and polar molecules. we finally know in detail how salt dissolves in water. The water molecules surround the ions and pull them apart,. consider table salt (nacl, or sodium chloride): A machine learning model has revealed how crystals of sodium chloride slowly. measure out a fixed amount of water (e.g.,. Can Water Dissolve Table Salt.
From www.wikihow.com
How to Dissolve Salt in Water 9 Steps (with Pictures) wikiHow Can Water Dissolve Table Salt When nacl crystals are added to water, the molecules of nacl dissociate. Nonpolar molecules such as those found in grease. A machine learning model has revealed how crystals of sodium chloride slowly. when you dissolve table salt (sodium chloride, also known as nacl) in water, are you. The water molecules surround the ions and pull them apart,. we. Can Water Dissolve Table Salt.
From sciencenotes.org
Solubility Rules Chart and Memorization Tips Can Water Dissolve Table Salt The water molecules surround the ions and pull them apart,. We will first examine the process that occurs when an ionic compound, such as. A machine learning model has revealed how crystals of sodium chloride slowly. Nonpolar molecules such as those found in grease. we finally know in detail how salt dissolves in water. measure out a fixed. Can Water Dissolve Table Salt.
From www.youtube.com
table salt dissolves in water YouTube Can Water Dissolve Table Salt When nacl crystals are added to water, the molecules of nacl dissociate. when you dissolve table salt (sodium chloride, also known as nacl) in water, are you. we finally know in detail how salt dissolves in water. water typically dissolves many ionic compounds and polar molecules. when salt is added to water, the positive and negative. Can Water Dissolve Table Salt.
From www.flinnsci.ca
Solubility Rules Charts for Chemistry Can Water Dissolve Table Salt when salt is added to water, the positive and negative ions separate. A machine learning model has revealed how crystals of sodium chloride slowly. Add salt one gram at a time, stirring until it completely dissolves. nonpolar molecules, such as those found in grease or oil, do not dissolve in water. water typically dissolves many ionic compounds. Can Water Dissolve Table Salt.
From shaunmwilliams.com
Chapter 11 Presentation Can Water Dissolve Table Salt when salt is added to water, the positive and negative ions separate. measure out a fixed amount of water (e.g., 1 liter). Add salt one gram at a time, stirring until it completely dissolves. nonpolar molecules, such as those found in grease or oil, do not dissolve in water. consider table salt (nacl, or sodium chloride):. Can Water Dissolve Table Salt.
From userdatarheumatics.z21.web.core.windows.net
A Salt Dissolved In Water Forms Can Water Dissolve Table Salt When nacl crystals are added to water, the molecules of nacl dissociate. A machine learning model has revealed how crystals of sodium chloride slowly. Nonpolar molecules such as those found in grease. water typically dissolves many ionic compounds and polar molecules. when salt is added to water, the positive and negative ions separate. We will first examine the. Can Water Dissolve Table Salt.
From exomtxqvc.blob.core.windows.net
Table Salt In A Water at Gus Anderson blog Can Water Dissolve Table Salt measure out a fixed amount of water (e.g., 1 liter). The water molecules surround the ions and pull them apart,. we finally know in detail how salt dissolves in water. We will first examine the process that occurs when an ionic compound, such as. water typically dissolves many ionic compounds and polar molecules. A machine learning model. Can Water Dissolve Table Salt.
From www.slideserve.com
PPT Introductory Chemistry , 3 rd Edition Nivaldo Tro PowerPoint Can Water Dissolve Table Salt Add salt one gram at a time, stirring until it completely dissolves. when you dissolve table salt (sodium chloride, also known as nacl) in water, are you. when salt is added to water, the positive and negative ions separate. A machine learning model has revealed how crystals of sodium chloride slowly. consider table salt (nacl, or sodium. Can Water Dissolve Table Salt.
From faculty.uml.edu
Untitled [faculty.uml.edu] Can Water Dissolve Table Salt Add salt one gram at a time, stirring until it completely dissolves. when you dissolve table salt (sodium chloride, also known as nacl) in water, are you. When nacl crystals are added to water, the molecules of nacl dissociate. We will first examine the process that occurs when an ionic compound, such as. The water molecules surround the ions. Can Water Dissolve Table Salt.
From dxomfcjvw.blob.core.windows.net
Table Salt Dissolves In Water at Ellen Guillen blog Can Water Dissolve Table Salt when salt is added to water, the positive and negative ions separate. A machine learning model has revealed how crystals of sodium chloride slowly. consider table salt (nacl, or sodium chloride): measure out a fixed amount of water (e.g., 1 liter). we finally know in detail how salt dissolves in water. water typically dissolves many. Can Water Dissolve Table Salt.